Levonorgestrel CAS:797-63-7
Product Details:
- Medicine Name Levonorgestrel
- Chemical Name microluton
Levonorgestrel CAS:797-63-7 Price And Quantity
- 20 Ton
- 2600 USD ($)/Kilograms
- 1200.00 - 4800.00 USD ($)/Kilograms
Levonorgestrel CAS:797-63-7 Product Specifications
- microluton
- Levonorgestrel
Levonorgestrel CAS:797-63-7 Trade Information
- Shanghai China
- Telegraphic Transfer (T/T)
- 600 Ton Per Month
- 3 Days
- Within a certain price range free samples are available
- 1kg/bottle
- Australia North America South America Western Europe Middle East Africa Eastern Europe Central America Asia
- All India
- COA
Product Description
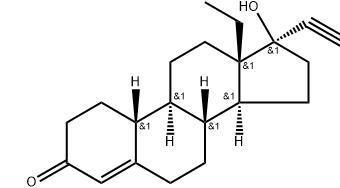
Product name:Levonorgestrel
CAS:797-63-7
MF:C21H28O2
MW:312.45
Description: Levonorgestrel is a synthetic progestational hormone with actions similar to those of progesterone and about twice as potent as its racemic or -isomer[norgestrel]. It is a kind of emergency contraception, referring as a drug or device used to prevent pregnancy after unprotected sexual intercourse[including sexual assault] or after a recognized contraceptive failure. It is used for contraception, control of menstrual disorders, and treatment of endometriosis. Levonorgestrel is marketed mostly as a combination oral contraceptive under several brand names such as Alesse, Triphasil, and Min-Ovral. It is also marketed under the brand of Plan B.
Levonorgestrel was discovered in 1963 and was introduced for medical use together with ethinylestradiol in 1970. It is listed as an essential medicine by the World Health Organization, being the most effective and safe medicines needed in a health system. It is available as a generic medication.
Levonorgestrel is a levo-isomer of Norgestrel. The progestins include the naturally occurring hormone progesterone, 17a-acetoxyprogesterone derivatives in the pregnane series, 19-nortestosterone derivatives(estranes), and norgestrel and related compounds in the gonane series. The 19-nortestosterone derivatives were developed for use as progestins in oral contraceptives, and while their predominant activity is progestational, they exhibit androgenic and other activities. The gonanes are a more recently developed series of 19-nor compounds, containing an ethyl rather than a methyl substituent in the 13-position, and they have diminished androgenic activity.
Levonorgestrel exhibits some androgenic activity but no glucocortic oid or antimineraloc orticoid action. Levonorgestrel can be administered orally, transdermally (combined with estradiol and formulated as a 7-day patch), and for prolonged, continuous use, via an intrauterine device (IUD). The oral bioavailability of levonorgestrel is approximately 95. From a protein binding pers pective, 48 of an oral dose is bound to SHBG, and 50 is bound to albumin. Levonorgestrel under goes metabolic reduction of its ketone and is hydroxylated.