Doripenem (S-4661)
Doripenem (S-4661) Price And Quantity
- 130.00 - 350.00 USD ($)/Gram
- 20 Ton
- 180 USD ($)/Gram
Doripenem (S-4661) Trade Information
- Shanghai China
- Telegraphic Transfer (T/T)
- 600 Ton Per Month
- 3 Days
- Within a certain price range free samples are available
- 10g/bottle
- Australia North America Central America South America Western Europe Asia
- All India
- COA
Product Description
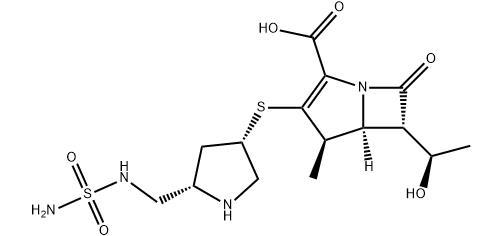
Product name:Doripenem(S-4661)
CAS:148016-81-3
MF:C15H24N4O6S2
MW:420.5
Description: Doripenem is a parenteral carbapenem antibiotic launched in Japan for the treatment of bacterial respiratory and urinary tract infections. It is a methyl carbapenem derivative, and it is the fourth analog to be marketed in this series following the launch of meropenem, biapenem, and ertapenem in previous years. The introduction of a 1-methyl group to the carbapenem skeleton enhances metabolic stability to renal dehydropeptidase-1 (DHP-1) and leads to improved antibacterial potency. The mechanism of action is likely to involve covalent modification of peptidoglycan biosynthetic enzymes responsible for catalyzing the final transpeptidation step of cell wall biosynthesis. The chemical synthesis of doripenem involves the coupling of a commercially available 4-nitrobenzyl protected 1 methylcarbapenem enolphosphate intermediate with a protected version of 2-(sulfamidomethyl)- 4-mercaptopyrrolidine as the key step. The requisite pyrrolidine intermediate is prepared in six steps starting from trans-4-hydroxy-L-proline. In vitro, doripenem exhibits activity similar to that of imipenem against Grampositive pathogens, and to that of meropenem against Gram-negative pathogens. The key differentiator for doripenem is its superior activity against Pseudomonas aeruginosa (MIC90=3.13 g/mL) as compared with meropenem and imipenem. Additionally, it possesses higher stability than imipenem or meropenem against mammalian dehydropeptidase I, and it is stable to most serine-based beta-lactamases. The route of administration of doripenem is either intravenous or intramuscular. It has low plasma protein binding (8.1%), and the primary route of elimination is renal.