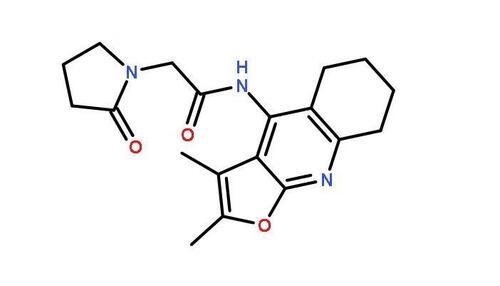
Leviitra CAS No.:224785-90-4
Product Details:
- Grade Industrial Grade
1600 USD ($)/Kilograms
X
Leviitra CAS No.:224785-90-4 Price And Quantity
- 20 Ton
- 1600 USD ($)/Kilograms
- 1000.00 - 3000.00 USD ($)/Kilograms
Leviitra CAS No.:224785-90-4 Product Specifications
- Industrial Grade
Leviitra CAS No.:224785-90-4 Trade Information
- Shanghai China
- Telegraphic Transfer (T/T)
- 300 Ton Per Month
- 3 Days
- Within a certain price range free samples are available
- 1kg/bottle
- Middle East Eastern Europe Africa Australia Central America North America South America Western Europe Asia
- All India
- COA
Product Description
Enter Buying Requirement Details