Cefoxitin Medicine
Cefoxitin Medicine Price And Quantity
- 165 USD ($)/Kilograms
- 20 Ton
- 120.00 - 330.00 USD ($)/Kilograms
Cefoxitin Medicine Trade Information
- Shanghai China
- Telegraphic Transfer (T/T)
- 600 Ton Per Month
- 3 Days
- Within a certain price range free samples are available
- 1kg/bottle
- Australia South America Western Europe Central America Asia North America
- All India
- COA
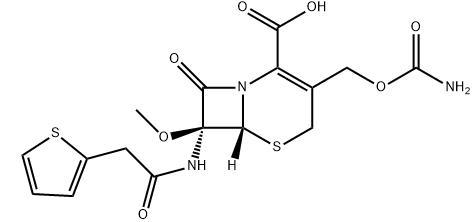
Product Description
Product name:Cefoxitin(Mefoxin)
CAS:35607-66-0
MF:C16H17N3O7S2
MW:427.45
Description: Cefoxitin contains the same C-7 side chain as cephalothin and the same C-3 side chain as cefuroxime. The most novel chemical feature of cefoxitin is the possession of an-oriented methoxyl group in place of the normal H-atom at C-7. This increased steric bulk conveys very significant stability against -lactamases. The inspiration for these functional groups was provided by the discovery of the naturally occurring antibiotic cephamycin C derived from fermentation of Streptomyces lactamdurans. Cephamycin C itself has not seen clinical use but, rather, has provided the structural clue that led to useful agents such as cefoxitin. Agents that contain this 7 methoxy group are commonly referred to as cephamycins. Ingenious chemical transformations now enable synthetic introduction of such a methoxy group into cephalosporins lacking this feature.





